Ansi asq z1 4 pdf11/11/2022  Squeglia available from ASQ has been widely adopted for this reason. Initially you start at normal inspection, and can move to either tightened or reduced inspection depending on how lots are dispositioned. Already Subscribed to this document.Īdd to Alert PDF. GLENDAY SIEVE PDF ANSI/ASQ Z1.4-2003 and ANSI/ASQ Z1.9-2003 Thus, the important step is to determine the AQL. It is important to understand what you are doing when using sampling plans, what they are and the protection you are trying to ensure. If it falls on an arrow does it mean that I have to change to the next sample size based on where the arrow points? Question I am confused about the values used for AQLs. 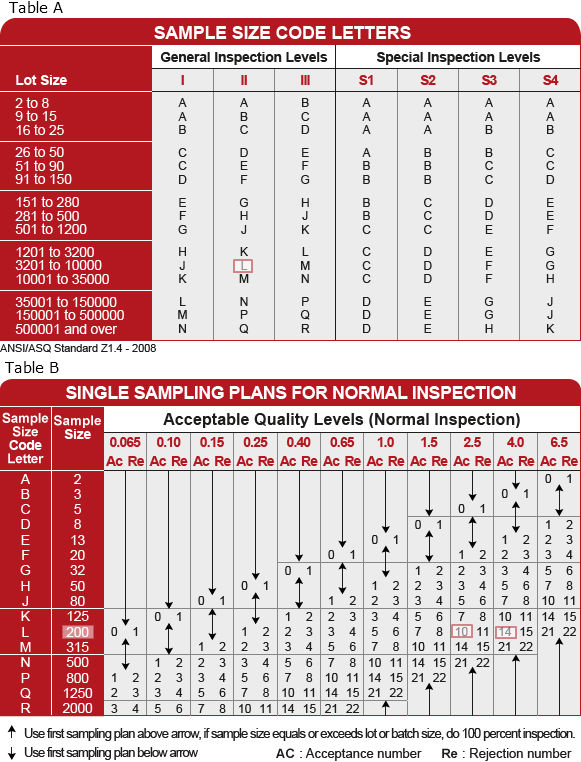
This is also what was intended by the creators of the sampling scheme. 
It is expected that the product quality level will be less than the AQL to avoid excessive non-accepted lots. Some interpret it to mean if a lot has AQL percent defective or less, a lot would have a high probability of being accepted based on the sampling plan. I can decide to chose the number of rolls to sample from using the tables given in Z1. This standard is also aqs to be included in Standards Subscriptions. Difference between ANSI/ASQC and ANSI/ASQ Need more than one copy? You can download and open this file to your own computer but DRM prevents opening this file on another computer, including a networked server. For more information about inspection, please view the resources found here. Source: Mil-Std E, replaced by commercial standards: ISO, ANSI/ASQ Z, NF, BS How to read the “ANSI tables”, aka “AQL tables”. ANSI/ASQ Z (R) Sampling Procedures and Tables for Inspection by Attributes Sampling Procedures and Tables for Inspection by Attributes is an. View Contents of Z1.ANSIASQZRSampling Procedures and Tables for Inspection by Attributes-Sampling Procedures and Tables for Inspection by Attributes is an. Identification and quantification of degradation products from polymeric medical devicesANSI/AAMI ST79:2006 and A1:2008- Comprehensive guide to steam sterilization and sterility assurance in health care facilities (Consolidated Text)ASTM F1886/F1886M-09- Standard Test Method for Determining Integrity of Seals for Flexible Packaging by Visual Inspection This product replaces:ASQ Z1.4-2003 and Z1.9-2003 SET - Sampling Procedures and Tables for Inspection by Attributes (Z1.4-2003) and Sampling Procedures and Tables for Inspection by Variables for Percent Nonconforming (Z1.9-2003) SETĪNSI/ASME Y14.5-2009- Dimensioning and TolerancingASTM F88/F88M-09- Standard Test Method for Seal Strength of Flexible Barrier MaterialsISO 9001:2008- Quality management systems - RequirementsASTM D5276-98(2009)- Standard Test Method for Drop Test of Loaded Containers by Free FallISO 17665-1:2006- Sterilization of health care products - Moist heat - Part 1: Requirements for the development, validation and routine control of a sterilization process for medical devicesISO 15223-1/Amd1:2008- Medical devices - Symbols to be used with medical device labels, labelling and information to be supplied - Part 1: General requirements - Amendment 1ASTM D4169-08- Standard Practice for Performance Testing of Shipping Containers and SystemsBS EN ISO 10993-13:2009- Biological evaluation of medical devices. This product referenced by:ANSI/AWS C3.11M/C3.11:2011 - Sampling Procedures and Tables for Inspection by Attributes (Z1.4-2008) and Sampling Procedures and Tables for Inspection by Variables for Percent Nonconforming (Z1.9-2008) SET This product includes:ANSI/ASQ Z1.4-2008 - Sampling Procedures and Tables for Inspection by AttributesĪNSI/ASQ Z1.9-2008 - Sampling Procedures and Tables for Inspection by Variables for Percent Nonconforming This product is related to: ASQ H1331 - Sampling Procedures and Tables for Inspection by Attributes (Z1.4-2008) and Sampling Procedures and Tables for Inspection by Variables for Percent Nonconforming (Z1.9-2008) SET It is applicable only when the normality of the measurements is assured. Variation may be measured by sample standard deviation, sample range, or known standard deviation. It provides tightened, normal, and reduced plans to be used on measurements which are normally distributed. Sampling Procedures and Tables for Inspection by Variables for Percent Nonconforming is an acceptance sampling system to be used on a continuing stream of lots for AQL specified. 
It provides tightened, normal, and reduced plans to be applied for attributes inspection for percent nonconforming or nonconformities per 100 units. Sampling Procedures and Tables for Inspection by Attributes is an acceptance sampling system to be used with switching rules on a continuing stream of lots for AQL specified. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
